Our FluoroSpot products
Published: November 1, 2023
Updated: June 24, 2025
6 minute read
Authored by: Tyler Sandberg
The principles of FluoroSpot
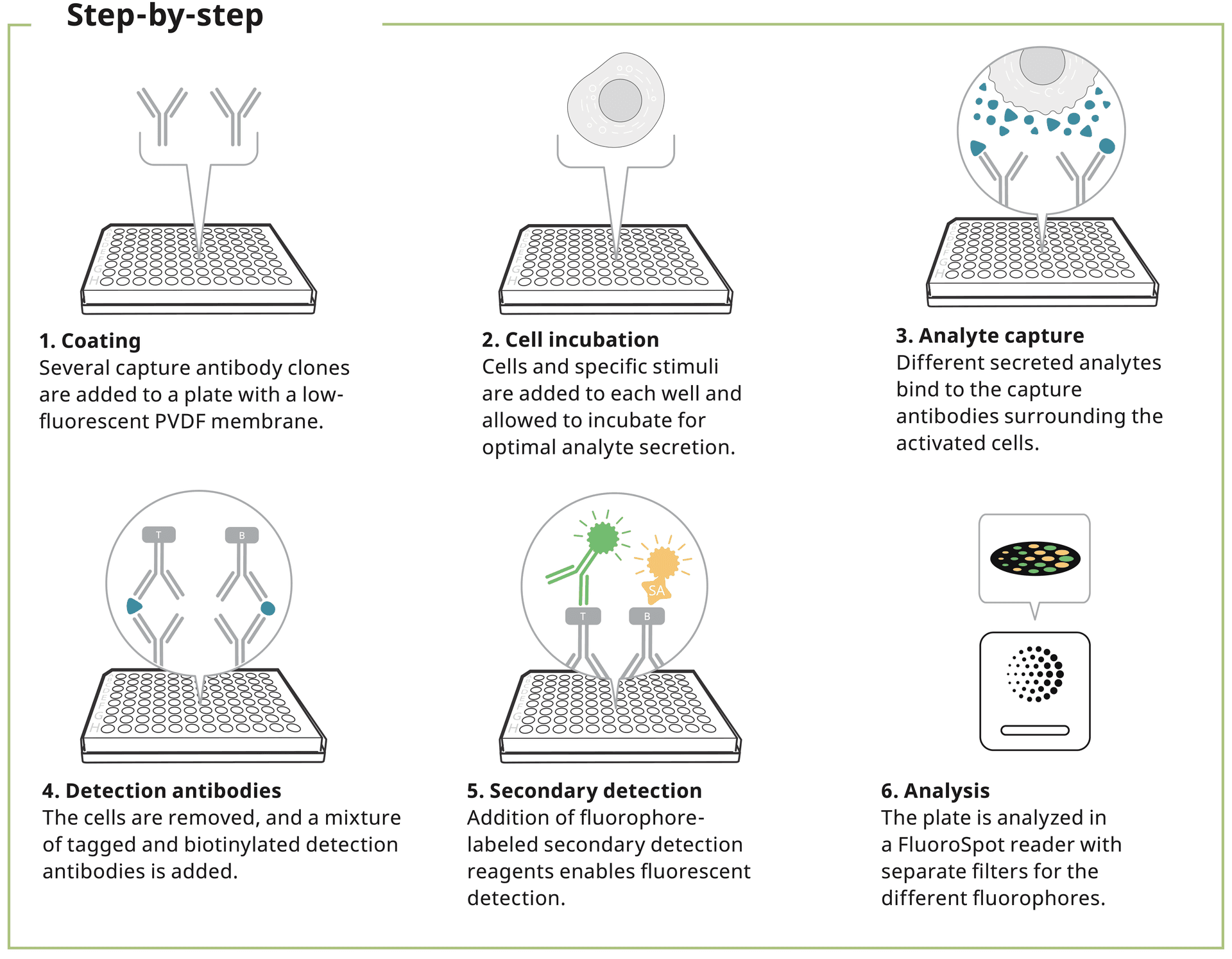
FluoroSpot is used to detect and quantify protein-secreting cells. This immunoassay combines the sensitivity of ELISpot with the capacity to study the secretion of several analytes simultaneously.
In FluoroSpot, a cell suspension is added to the wells of a plate coated with one or more protein-specific capture antibodies. Proteins secreted by the cells are immediately captured by these antibodies during the entire stimulation period. The cells are removed and then tagged or biotinylated protein-specific detection antibodies are added followed by fluorophore-labeled secondary reagents.
The plate is then read using a FluoroSpot reader where each fluorescent spot corresponds to a responding analyte-secreting cell.
What are the benefits of FluoroSpot?
Identify rare cells
FluoroSpot is one of the most sensitive cellular assays available, up to 500 times more sensitive than intracellular cytokine staining. If one cell secretes the analyte, it’s detected and visualized as a single spot.
Study analytes with different kinetics
With FluoroSpot, the release of multiple cytokines or immunoglobulins can be studied at the single-cell level. Cytokines released directly after activation can be analyzed together with cytokines that have slower kinetics without manipulating intracellular processes.
Get the most out of your samples
FluoroSpot's multiplexing capacity allows for the simultaneous detection of various cytokines from a single cell, yielding a wealth of data without the need for a larger cell sample. With B cell FluoroSpot, you can analyze multiple immunoglobulins or antigen-specific B cells in the same well. This efficiency maximizes the use of limited or precious samples, enabling more comprehensive data analysis from your cells. As a result, researchers can perform more detailed and advanced immunological assessments, even when cell quantities are restricted.
Extreme sensitivity
To compare the sensitivity of FluoroSpot with flow cytometry, increasing numbers of transfected CHO cells constitutively secreting IFN-γ and IL-10 were mixed with 105 non-transfected cells, shown on the x-axis. The number of spot-forming cells (SFC) is depicted on the left y-axis and the frequency of cells stained intracellularly for cytokine (ICS) on the right y-axis.
FluoroSpot could detect cytokine secretion when only 10 transfected cells were added. By contrast, at least 5,000 transfected cells were required to detect the cytokines by flow cytometry. (Figure adapted from Chauvat et al., Hum Vaccin Immunother: 2014).
FluoroSpot 500x more sensitive than ICS
Increasing numbers of transfected cells were mixed with non-transfected cells.
FluoroSpot applications and usage
Due to its high sensitivity, FluoroSpot is ideal for identifying polyfunctional cells in e.g., vaccine or cancer research. The detection of different secretory profiles separates the responding cells into different functional groups. Looking at several proteins in parallel increases the possibility of identifying distinct cell populations in the same well.
IFN-γ is often combined with IL-2, TNF-α, or Granzyme B in analyses investigating cytotoxic T cell responses against selected antigens. Stimulation is usually achieved with peptide pools covering the vaccine antigen. Check out all our FluoroSpot combinations here.
A 3-color FluoroSpot assay can be used to identify up to 7 secretory profiles from single- to double- even triple-secreting cells.
FluoroSpot analysis showing three individual images from the same well and an image overlay, combining images from the three filters.
Polyfunctional T cell profiling
FluoroSpot's advanced multiplexing allows for the detailed analysis of secretory profiles within individual immune cells, providing key insights into immune system dynamics, such as Th1 or Th2 responses, or cytotoxic T lymphocyte activity. This intricate detection of multiple cytokines enables a more nuanced understanding of immune cell function and the orchestration of immune responses, crucial for refining strategies in vaccine research and the development of novel immunotherapies. The assay can facilitate a deeper characterization of the immune response, leading to enhanced immune profiling.
Different secretory profiles provide insights into the CTL or T helper cell response.
Enhanced B cell profiling
FluoroSpot excels in B cell profiling by capturing multiple immunoglobulin classes or isotypes directly upon secretion, enhancing the detection of specific antibody responses. The assay not only makes it easy to identify rare antigen-specific antibody-secreting cells (ASCs) with high sensitivity but also allows for the assessment of circulating memory B cells. Regularly employed in immunological research, B cell FluoroSpot is particularly valuable for analyzing the breadth and specificity of B cell responses following infection or vaccination. Learn more about B cell profiling here.
B cell ELISpot and FluoroSpot can be used to measure both acute and memory B cell responses. Both total and antigen-specific B cells can be enumerated.
Characterize vaccine-induced immunity
Like ELISpot, FluoroSpot is useful in assessing vaccine-induced immunity by quantifying antigen-specific T and B cell responses but giving a broader picture of the response by enumerating highly functional triple-secreting IFN-γ/IL-2/TNF-α cells or even multiple immunoglobulins in the same well. The assays allow researchers to gauge a vaccine's immunogenicity and potential protective effects, providing critical insights into their performance and function. Our white paper takes a deeper look into ELISpot's role in vaccine development, offering a detailed perspective on its application in shaping vaccine-mediated immunity.
Cancer and immunotherapy
Mabtech's FluoroSpot kits enable precise immune monitoring, capturing the activity of killer T cells through key markers like IFN-γ and Granzyme B simultaneously. This technology has been instrumental in advancing cancer immunotherapy research, offering insights into how the immune system targets and destroys cancer cells. Explore our white paper to see how FluoroSpot has facilitated this groundbreaking work, spotlighting the assay's critical role in the development of novel cancer treatments.
Our kit formats
Flexible, validated, or down a charted path – it’s your choice. With FluoroSpot Flex you can select analytes and create over 50,000 possible combinations. FluoroSpot Plus kits have validated analyte combinations and pre-coated plates to save time and minimize variability. Finally, FluoroSpot Path kits include specific antigens and are designed to study immune responses to a specific pathogen.
The future of FluoroSpot analysis
Because the assay principle is similar to ELISpot, if one cell secretes the analyte, it is detected and visualized as one spot. However, whereas spots in ELISpot can be seen with your naked eye, fluorescent spots need to be analyzed in an automated reader equipped with an exciting light source and suitable emission filters. In addition, the reader needs a spot-counting algorithm capable of identifying spot centers and creating an overlay analysis. Our reader Mabtech IRIS™ 2 is optimized for FluoroSpot analysis.
IRIS 2 works seamlessly with the fluorophores in our FluoroSpot kits equipped with filters for the following wavelengths (excitation/emission):
-
380 nm/430 nm (LED380, DAPI)
-
490 nm/510 nm (LED490, FITC)
-
550 nm/570 nm (LED550, Cy3)
-
640 nm/660 nm (LED640, Cy5)
The novel software is optimized for precise spot center detection ensuring accurate multiplexing and also introduces the newest FluoroSpot data, relative spot volume. Book an online demo!
To get the most out of your assay, download our Step-by-step guide to FluoroSpot.
Selected references
Our FluoroSpot kits appear in numerous publications ranging from vaccine development to cancer research. Check out our Publication database for a full list of references.
Jahnmatz et al., Memory B-Cell Responses Against Merozoite Antigens After Acute Plasmodium falciparum Malaria, Assessed Over One Year Using a Novel Multiplexed FluoroSpot Assay, Front Immunol. 2020
Singhania et al. The TCR repertoire of alpha- alpha-synuclein-specific T cells in Parkinson’s disease is surprisingly diverse, Sci Rep. 2021
Bronge et al., Myelin oligodendrocyte glycoprotein revisited-sensitive detection of MOG-specific T-cells in multiple sclerosis, Journal of autoimmunity 2019
Mateus et al., Selective and cross-reactive SARS- CoV-2 T cell epitopes in unexposed humans, Science 2020
Ishigaki et al., No Tumorigenicity of Allogeneic Induced Pluripotent Stem Cells in Major Histocompatibility Complex-matched Cynomolgus Macaques, Cell Transplant. 2021