Streamlined workflow
An overlooked time-consuming aspect of ELISpot was data analysis. With pre-calibrated settings and handy analysis tools, the time from data acquisition to ready-made analysis has been reduced to a minimum.
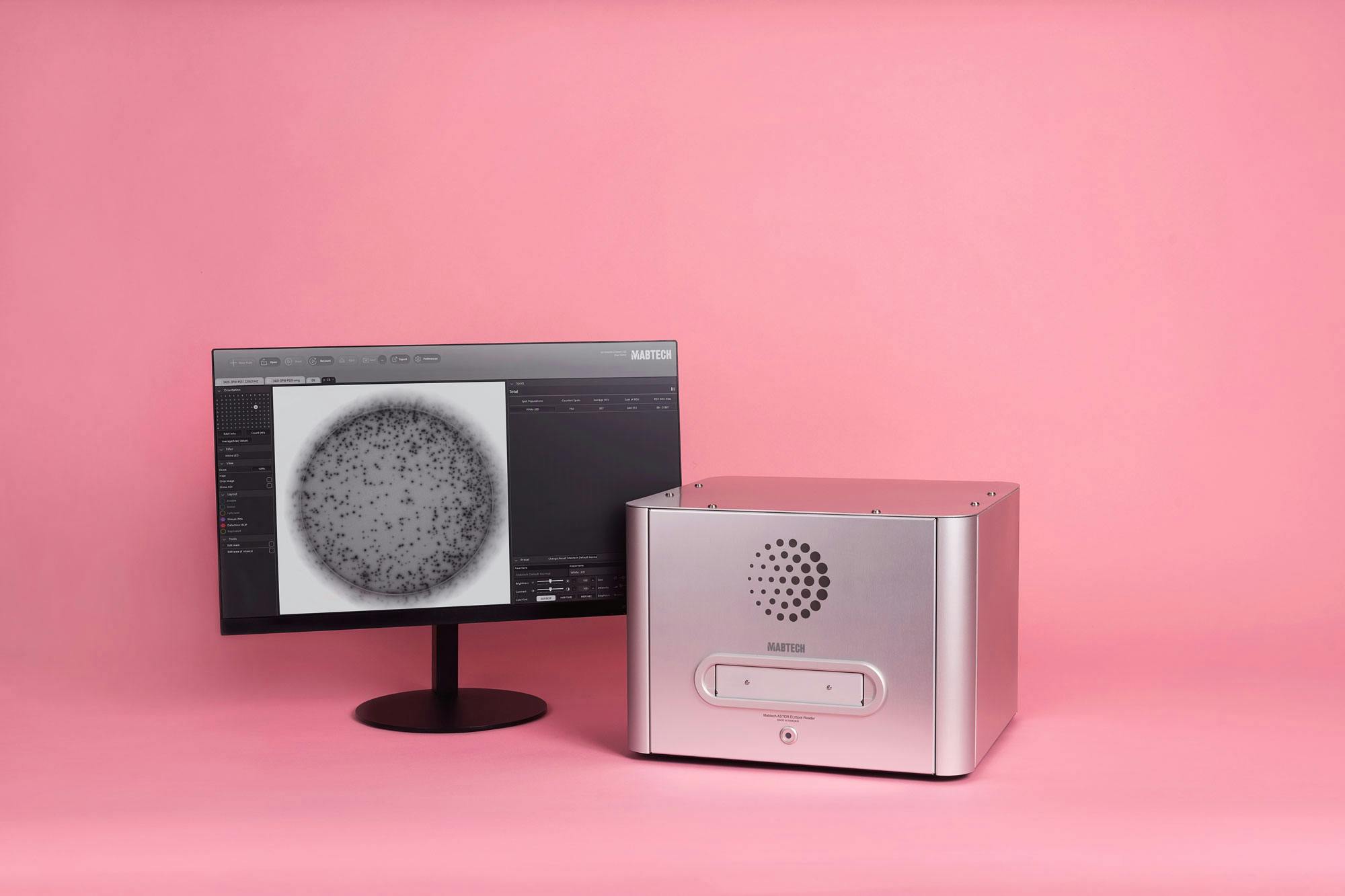
Precise spot counting
The software for Mabtech ASTOR is based on our patented RAWspot™ technology. The refined spot center localization lays the foundation for accurate spot counting. This can be confirmed, for example, by how linear the counting is in correlation to a cell dilution. Mabtech ASTOR provides linear spot counting with more than 3,000 spots per well, allowing correct spot processing for a multitude of setups including positive controls.
Read now, analyze later
Select default settings for quick data acquisition. Preferred spot size, plate layout, and image setups can be easily applied after plate reading, without affecting the original data. Find out how Mabtech ASTOR meets the requirements of CFR21 part 11 (FDA guidelines on electronic records in clinical investigations).
Ready for automation
Mabtech ASTOR 2 was designed to make the analysis of larger projects as painless as possible. In addition to self-calibration, responsive software, and easy data handling, ASTOR 2 allows for robotic plate loading. Our reader can be part of an automated workflow with a robotic arm or be fully integrated into a larger automated setup. Each automation solution is unique, and we’ll be here to help every step of the way.
Performance qualification
Our readers are reliable tools that can be trusted to deliver accurate results over an extended period of use. However, it’s essential to demonstrate that the readers maintain acceptable performance over time, especially when facing internal or external review for regulatory compliance. Mabtech’s Performance Qualification (PQ) plate provides a solution for verifying that our instruments fulfill quality specifications and to ensure reproducibility. The PQ plate allows users to conduct a series of quantitative and qualitative tests, making it easy to identify any incorrect or inconsistent operation of the reader. By using PQ plates, our instruments can be confidently validated, providing peace of mind knowing that experiments are accurate and reliable.
Different sections of the PQ-plate for ASTOR:
1. Tests focus and RAWspot algorithm in ELISpot
2. Tests white LED light intensity in ELISpot