Step-by-step guide to FociSpot
Published: October 12, 2023
Updated: October 3, 2024
4 minute read
Authored by: Tyler Sandberg
Learn the basics of our newest assay platform, FociSpot, and get ready to detect virus-infected cells. Here, we'll guide you through each step to make sure you get the best results possible.
What is FociSpot?
FociSpot is our new assay platform that includes both kits and a brand-new analysis capability of Mabtech IRIS™ 2 allowing for automated counting of foci in a 96-well plate format. Focus forming assays (FFAs) can have a variety of different setups for different research goals such as focus-reduction neutralization tests (FRNT) or Tissue Culture Infectious Dose (TCID50) assays. FRNT and TCID50 can be used to quantify viral titers or virus-specific antibody neutralization titers and are commonly used in vaccine and infection research. We know that each viral assay is unique, using different cell lines, media, and viruses – but counting foci doesn’t have to be. In these assays, foci are detected with immunostaining using virus-specific mAbs and a precipitating substrate reaction similar to ELISpot. The developed foci can be easily counted using the FociSpot application in IRIS 2.
An example of a typical FRNT assay using FociSpot to enumerate neutralizing antibody titers in a given sample.
Assay principle
In FociSpot, virus foci are detected by using mAbs specific to viral proteins. After fixation and permeabilization of infected cells in a 96-well plate, the virus-specific biotinylated detection mAb is added. The use of Streptavidin-ALP enzyme conjugate, followed by a precipitating substrate enables detection of foci. Foci are preferably counted with Mabtech IRIS 2, which offers high-throughput counting and easy data handling. Foci can also be counted manually using a microscope.
Important to note:
-
The protocol for virus infection of cells needs to be established by the user prior to starting the FociSpot assay.
-
Consult local regulations for handling of virus and cells, considering biological hazards and waste handling.
-
Formaldehyde and methanol used for fixation and permeabilization are chemicals with hazard classifications. Consult safety data sheets from the manufacturer and follow local regulations.
Below is an example step-by-step guide for measuring neutralizing antibody titers using FRNT50 (50% foci reduction neutralization test). Please note that FociSpot can be applied to measuring viral titers, infectious doses, and more! Additionally, different viruses will require different incubation times, susceptible cell lines, and altered protocols. Please have these steps established prior to running the FociSpot assay.
Procedure
1. Incubate virus and sample
-
Heat inactivate the sample serum at 56° C for 30 minutes either in a water bath or heat block.
-
In a separate 96-well plate, make serial dilutions of the test serum samples. Common dilutions are 1:10, 1:20, 1:40, and so on. Duplicates or triplicates are recommended.
-
Dilute the virus of interest to the appropriate FFU/ml in serum-free media and add to the serum dilution plate at a ratio of 1:1. Incubate for 1 hour at 37° C.
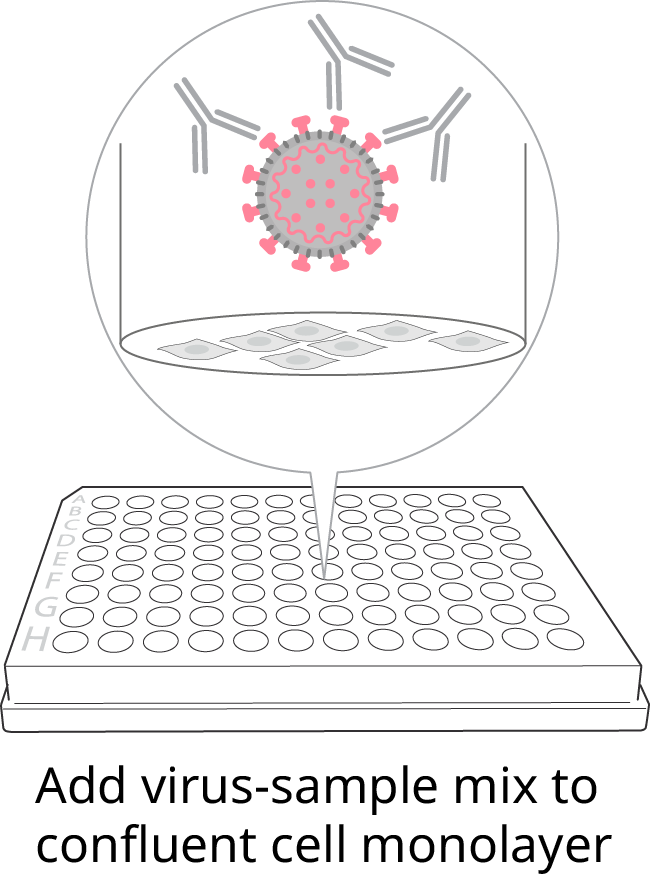
2. Titer onto confluent cells
-
Remove the medium from a prepared 96-well FociSpot plate with a healthy confluent cell monolayer and add the virus-serum mixture. Incubate 22-24 hours at 37° C.
-
Remove the virus-serum mixture from the plates and wash with PBS.
-
Fix the cells with a suitable fixative such as 80% acetone, 4% formaldehyde, or methanol.
3. FociSpot protocol – staining of infected cells
-
Remove the fixative and wash the plate 4 times with PBS.
-
If cells were fixed using formaldehyde, ice-cold methanol should be added to permeabilize the cells and incubated for 10 minutes at room temperature.
-
Wash the plate 4 times with PBS.
-
Block the plate with 1% BSA in PBS for one hour at room temperature.
-
Remove blocking and add the appropriate concentration of the detection mAb in PBS with 0.1% BSA. Incubate for 1 hour at room temperature.
-
Wash as above and add the Streptavidin-ALP diluted 1:1000 in PBS with 0.1% BSA. Incubate for 1 hour at room temperature.
-
Wash as above and add filtered BCIP/NBT-plus substrate. Develop until distinct foci emerge (5-30 minutes).
-
The development is stopped by thorough washing with tap water.
4. Foci analysis
-
Let the plates dry completely, preferably in the dark.
-
Read the plate using Mabtech IRIS 2.
In FRNT, the results are usually presented as a neutralization titer. This is defined as the reciprocal of the highest serum dilution that causes a specific percentage reduction (often 50%, hence FRNT50) in the number of foci compared to a control, typically a virus-only control.
In this image, Influenza A foci were detected with immunostaining using virus-specific mAbs.
Check out all of our FociSpot kits here. Is there a virus-specific mAb you're looking for? Let us develop it for you!