How to detect immunoglobulins with ELISA
Published: November 28, 2022
Updated: April 2, 2025
4 minute read
Authored by: Lara Mentlein
With ELISA, it's easy to detect and quantify immunoglobulins in solution, whether you're looking for antigen-specific or total Ig, for example, IgG, IgA... In this guide, we'll walk you through different assay setups, preparing you to tackle your specific research question.
Detecting antibodies with antibodies
Antibodies are proteins that specifically bind to a target molecule and, as such, are central players in the immune system of vertebrates. Over the last century, we humans have learned to utilize their unique characteristics for applications in the lab. Ultimately, we can use antibodies in assays to detect other antibodies.
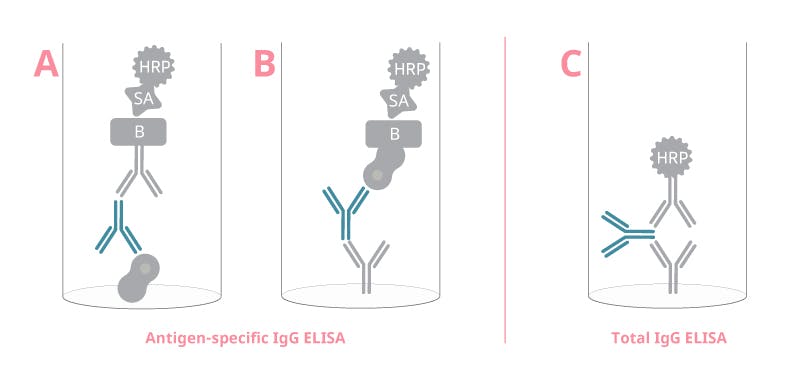
Figure 1. Assay principle of three different setups to detect IgG in ELISA. Antigen-specific IgG can be detected with either a A) indirect or B) reverse setup, and total IgG can be detected in a C) sandwich setup. This is an example of IgG evaluation; the assay can be modified to detect other Ig isotypes or subclasses as well, using the corresponding assay antibodies.
Antigen-specific immunoglobulins
The setup for detecting antigen-specific immunoglobulins differs from the classic sandwich assay. The most common method is to coat the antigen onto the plate first (Figure 1A). Alternatively, a tagged antigen can be used in a reverse setup (Figure 1B). Independently of which setup you choose, keep in mind that antigen-specific ELISAs heavily depend on antigens being in their native form or as close to it as possible. The antigens for the assay are usually recombinantly produced by researchers themselves, but some are also commercially available.
The typical setup for an antigen-specific Ig ELISA (Figure 2) includes these steps:
-
The antigen of interest is coated on the ELISA plate utilizing the hydrophobic binding of the antigen to the plastic. We recommend using a high-binding ELISA plate, for example, an ELISA plate made from polystyrene.
If you are setting up an entirely new assay, please be aware that the relevant epitope on the antigen might be hidden after the coating step. If you suspect hidden epitopes as the reason behind a non-working ELISA, you could try a different coating protocol. For example, you could test different coating buffers, such as PBS 7.4 or bicarbonate buffer pH 9.5, as the pH influences the hydrophobic binding between amino acid side chains and polystyrene. Alternatively, if you have the antigen available in a tagged format, you could test a reverse ELISA setup (Figure 1C).
-
Plate coating is followed by a blocking step.
-
After adding the samples, antigen-specific immunoglobulins should bind to the antigen coated on the plate.
-
Unbound immunoglobulins will be washed away in the subsequent washing steps.
-
The remaining antigen-specific immunoglobulins are detected with antibodies specific to the Fc portion of the bound immunoglobulins. This is often performed in an isotype-specific manner, for instance, IgG-only detection. Seen from a more practical perspective, either a one-step (Figure 2A) or a two-step detection setup (Figure 2B) can be used. As antigen-specific immunoglobulins may be low in abundance, a more sensitive two-step detection might be more suitable to amplify the detection signal.
Figure 2. Antigen-specific immunoglobulin ELISA using A) a direct ELISA with one-step detection or B) an indirect ELISA with two-step detection.
Applications for antigen-specific immunoglobulin detection
- Immune response to vaccines, infections, autoantigens, allergens, and more!
- Hybridoma screening
Isotype- or subclass-specific immunoglobulins
A typical sandwich assay setup can be used to detect antibodies of the same isotype (also called class) or of the same antibody subclass (Figure 1C, Figure 3). For this, you need a matching antibody pair specific for the Ig Fc portion (the antibody backbone) that differentiates the isotypes or subclasses. After coating the capture antibody to the ELISA plate, the Ig from the sample will be captured by the specific coating mAb(s) and then sandwiched by the detection antibody in the following step. As Ig isotypes are typically abundant in blood samples, a one-step detection setup (Figure 3A) might be preferred over two-step detection (Figure 3B) since signal amplification shouldn’t be needed. An exception to this is IgE, which should be analyzed with a two-step ELISA because of its low abundance in humans.
Figure 3. Total immunoglobulin isotype or subclass detection with a sandwich ELISA using either A) one-step or B) two-step detection.
Applications for anti-immunoglobulin ELISA
- Quantify total immunoglobulin levels in samples
- Reveal details of immune responses by determining immunoglobulin isotypes and subclasses
How to use Ig ELISA? – Here's an example
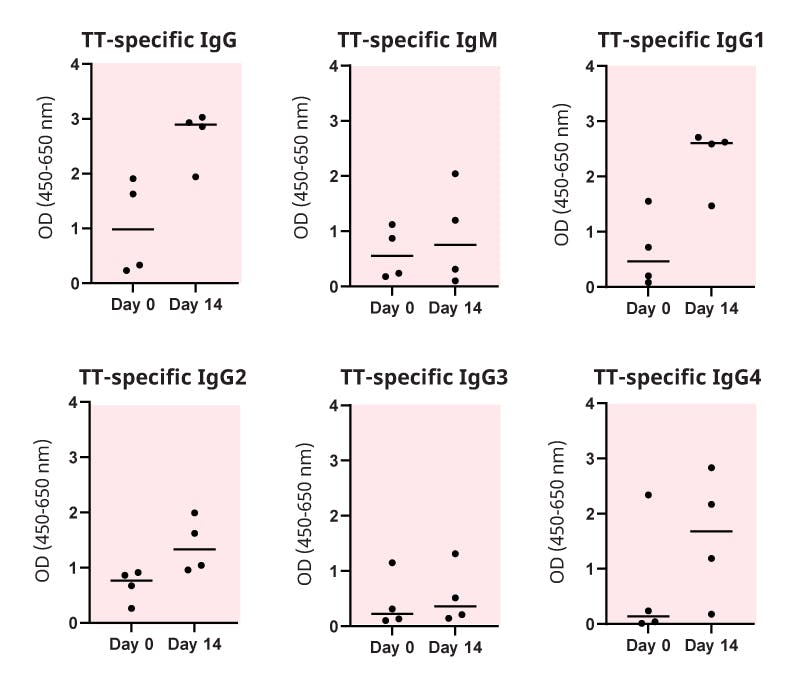
Figure 4. An example of the application of an antigen-specific Ig ELISA. Tetanus toxoid-(TT)specific Ig was measured with ELISA before and after booster admission. Plasma samples from four different donors were collected before and at two weeks after one booster dose of Covaxis (a combined vaccine including TT). The plasma samples were diluted 10,000x (IgG, IgM), 1,000x (IgG1), or 100x (IgG2, IgG3, IgG4), and the binding of TT-specific Ig was detected with isotype- or subclass-specific detection antibodies. (In-house data, Kesa et al.)
Tip
Pay attention to the differences in the ELISA protocols in case you combine isotype detection with antigen-specific detection on the same plate.